
The demand for reliable remote patient monitoring technology has never been greater. With hospitals under pressure to improve patient safety, reduce unplanned ICU transfers, and optimize staff efficiency, AI-driven solutions are shaping the future of care delivery. Among these global health innovations, Dozee stands out—India’s first contactless monitoring solution to receive U.S. FDA 510(k) clearance as a Class II medical device.
For healthcare leaders, this milestone is more than a regulatory approval. It signifies global validation of Dozee’s clinical accuracy and its ability to strengthen care delivery across diverse hospital ecosystems.
What is Dozee and What Makes It Unique?
Dozee is India’s pioneering AI-powered medical device for continuous, contactless monitoring. Installed under the mattress, the sensor captures micro-vibrations from cardiac contractions, respiration, and body movements—converting them into vital parameters like HR, RR, SpO₂, BP, and sleep cycles.
What makes Dozee unique:
- Contactless monitoring ensures patient comfort.
- AI-powered Early Warning System (EWS) detects deterioration 8–16 hours in advance.
- Scalable from ICUs to general wards, creating a continuum of monitoring.
- Proven in 300+ hospitals and 20,000+ beds, saving ~2.5 hours of nursing time daily per HCP.
What FDA 510(k) Clearance Means
For hospitals and providers, FDA clearance is an assurance of safety, reliability, and clinical effectiveness.
- 510(k) clearance confirms Dozee is substantially equivalent to established U.S. devices.
- Ensures compliance with stringent performance, quality, and safety benchmarks.
- Strengthens trust for both domestic adoption and international scaling.
For administrators, this clearance signals that Dozee’s remote patient monitoring devices meet global clinical standards while being designed and manufactured in India.
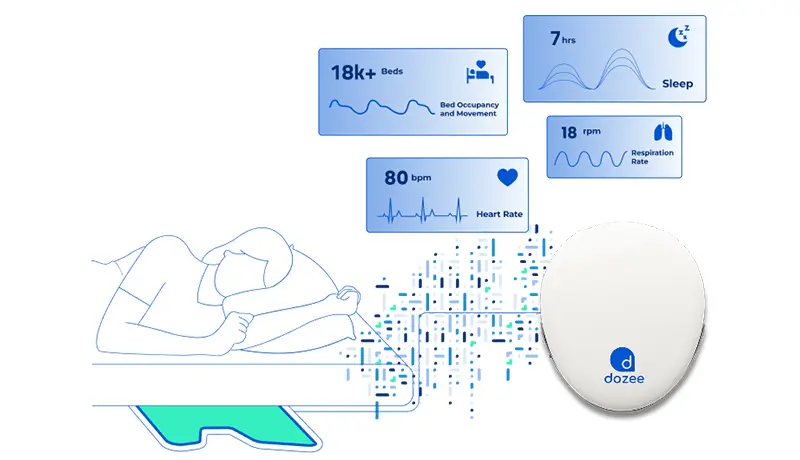
Core Features of Dozee’s RPM Tech
Dozee’s FDA-cleared RPM tech combines multiple innovations into one platform:
- Contactless sensor: placed under the mattress, requiring no patient wires.
- AI-powered analytics: continuous calculation of EWS, NEWS2, and MEWS.
- Smart alerts: customizable thresholds reduce alarm fatigue by 15–20x.
- Interoperability: seamless integration with HIS/EHR systems.
- SecureRPM: ISO 27001:2022, HIPAA, and SOC2 compliant for data protection.
How It Benefits Hospitals and Providers
Hospitals adopting Dozee’s remote patient monitoring devices have reported:
- 50% reduction in unplanned ICU admissions.
- 83% reduction in time spent on vitals checks.
- Improved nurse efficiency: ~2.5 hours saved daily per staff.
- Better patient outcomes: up to 144 lives saved annually per 100 beds.
For providers, this translates into more time for direct care and less administrative burden. For administrators, it means cost savings, compliance readiness, and stronger hospital reputation.
Why It’s Ideal for Healthcare Ecosystem
Dozee aligns with both Indian healthcare needs and global digital health trends:
- Resource-limited hospitals: quick 15-minute setup makes it feasible for Tier 2 and Tier 3 facilities.
- Tertiary care centers: scalable across wards for proactive safety and accreditation readiness.
- Telemedicine providers: enables remote triage and escalation.
- Global adoption: FDA clearance expands its applicability across international markets.
In a system where 90–95% of beds remain under-monitored, Dozee bridges the gap by converting every hospital bed into a smart monitoring hub.
Conclusion
Dozee’s FDA-cleared remote patient monitoring technology is more than a product—it is a validated, scalable, and patient-friendly pathway to safer hospitals. For administrators, clinicians, and health-tech leaders, it delivers a balance of global compliance and local relevance.
By enabling earlier detection, reducing nurse workload, and digitizing ward monitoring, Dozee is shaping the future of AI powered medical devices and setting new benchmarks in patient safety for India and the world.
FAQs
What does FDA cleared technology mean?
FDA clearance (via the 510(k) process) means a device has been reviewed by the U.S. Food and Drug Administration and found to be substantially equivalent in safety and effectiveness to an already legally marketed device. For hospitals, it signals reliability and clinical validation.
Is FDA cleared good?
Yes. FDA clearance is a strong indicator that the technology meets international quality, safety, and performance benchmarks. For hospital administrators, this provides confidence in procurement decisions, especially for AI powered medical devices like Dozee.
What is the full form of FDA in technology?
FDA stands for the Food and Drug Administration—the U.S. regulatory body responsible for protecting public health through oversight of food, drugs, and medical devices.
How do I find FDA-cleared devices? Hospitals and clinicians can search the FDA 510(k) Premarket Notification Database, which lists all cleared medical devices. Dozee’s inclusion in this registry reinforces its position as one of India’s first contactless remote patient monitoring devices with U.S. validation.
What is RPM technology in healthcare?
Remote patient monitoring technology (RPM) uses sensors, connectivity, and analytics to track patient vitals outside the ICU, often in general wards or even at home. Dozee’s contactless system captures continuous data (HR, RR, SpO₂, BP, sleep) and applies AI for early warning alerts, helping hospitals improve safety, efficiency, and outcomes.


